Pharmaceutical and Medical Device Security Program
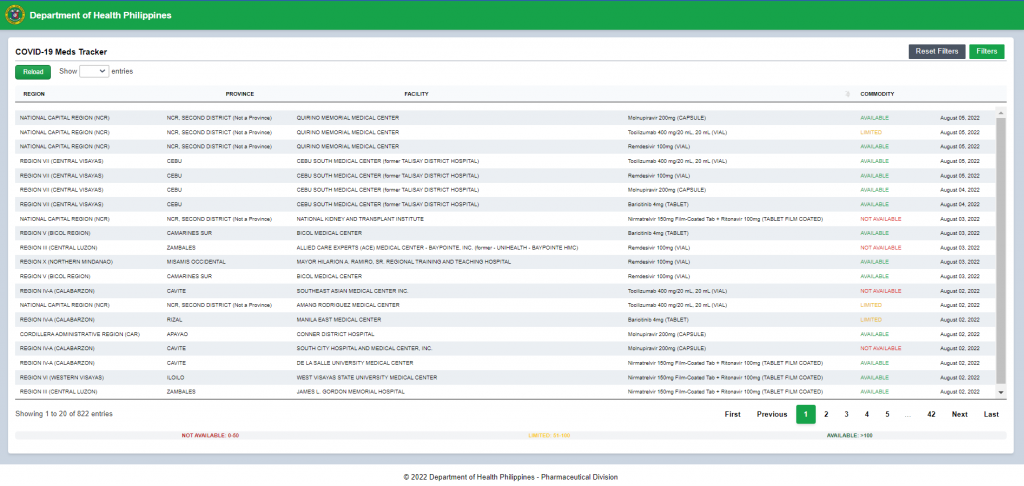
In virtue of the Department Personnel Order No. 2020 – 1032 entitled “Creation of a Technical Working Group (TWG) for the Formulation and Implementation of the Pharmaceutical and Medical Device Security Policy and Plans in light of COVID-19 Pandemic and other instances of Drug Shortages”, a TWG was created to formulate and implement the Pharmaceutical and Medical Devices Security Plan based on a comprehensive strategic framework that details the various health systems, policies, programs, financing requirements, and contextual environments that need to be in place to strengthen supplies for specific categories of medical devices as well as medicines. The Pharmaceutical Division (PD) was assigned to be the TWG secretariat. Likewise, PD was also tasked by the Response Cluster to monitor the supply of COVID-19 therapies in the hospitals, help address issues of drug shortages and coordinate with the Philippine Food and Drug Administration (FDA) on matters related to the registration of COVID-19 investigational drugs.
Drug Security Report
Frequently asked questions
Pharmaceutical and Medical Device Security is based on the presumption that uninterrupted sustainable access to life-saving essential pharmaceutical products and devices exists at all times in every health facility at an affordable price
Formulate and recommend policy guidelines for the establishment of a Pharmaceutical Security and Continuity Plan in the Country by ensuring a continuous and uninterrupted supply of essential medicines
- Monitoring of supply & consumption of COVID-19 therapies in the hospitals
- Coordination with the industry on their status of supply and the products that are at high risk/critical levels
- Coordination with the Food and Drug Administration (FDA) regarding concerns such as registration of COVID-19 investigational drugs & Drug Shortages
- Monitoring of prices of essential medicines & devices
- Setting Suggested Retail Price/Maximum Drug Retail Price
- Assisting the Department of Health (DOH) with regards to local and foreign donations
Related Issuances
Contact Us:
Telephone Number: 02-8757734 Loc. 253
Address: 4th Floor, Philippine Blood Center, Lung Center Compound, Quezon Avenue, Quezon City
Email Address: dohncpam@gmail.com